|
4/29/2023 0 Comments Hydrogen bonds in dna 
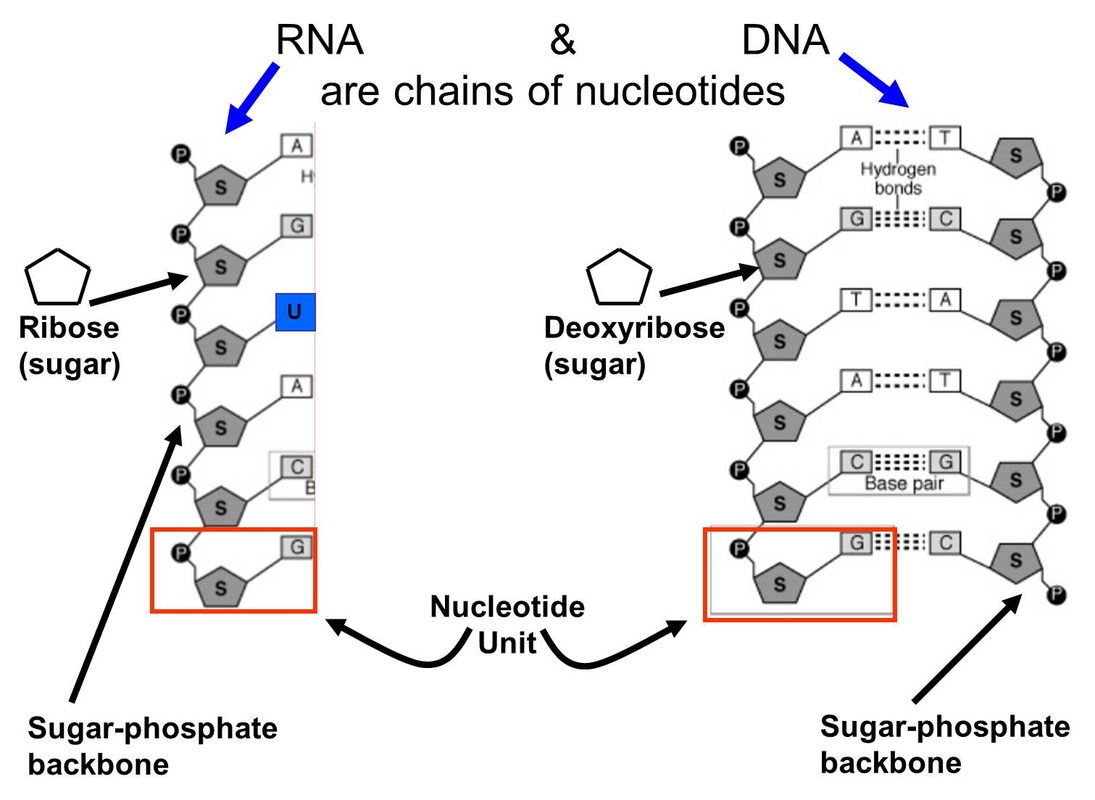
a deoxyribose sugar (pentose), a nitrogenous base, and a phosphate group.Ī large number of nucleotides bond together to form one strand of DNA. The two strands of DNA are composed of nucleotides and each nucleotide is a combination of three molecules viz. Let us study the bondings in DNA in detail.ĭNA is a double-stranded structure that serves as a carrier of biological information for most organisms. Therefore, multiple hydrogen bonds exist between the complementary nucleotides located on opposite strands of DNA. The adenine molecule forms a double bond with thymine while guanine forms a triple bond with cytosine. The purines always bond with pyrimidines, however, the number of bonds differs between the two types of base pairing. So, does DNA have hydrogen bonds? Yes, the two strands of the DNA molecules are joined through hydrogen bonds. However, another type of DNA called mitochondrial DNA which is maternally inherited is located in the mitochondria of the cell.Īn interesting question regarding bonds inside the DNA molecule is whether DNA consists of hydrogens bonds in it or not. Most DNA is located inside the nucleus of a cell. The different individuals, even when they belong to the same species do not have identical DNA except for the congenital twins that are born with the same DNA.ĭue to its uniqueness DNA also serves as a source of identification. It is responsible for the transmission of various traits from one generation to another.Įvery cell of the body contains its own DNA which is unique to every individual. Hydrogen bonds have a strength that is halfway between weak van der Waals forces and strong covalent bonds.Deoxyribonucleic acid, abbreviated as DNA is the hereditary material of almost all the living beings on earth. Strength of Hydrogen Bond: The hydrogen bond is a relatively weak one. The shared pair of electrons are attracted to the oxygen atoms more, and this end of the molecule becomes negative, while the hydrogen atoms become positive. A highly electronegative oxygen atom is connected to a hydrogen atom in the water molecule. Such compounds have molecular weights that are twice as large as those calculated from their simple formula.Īn excellent example of Hydrogen Bonding is Water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
